PDG WORKING PROCEDURES
General
Harmonization may be carried out retrospectively for existing monographs or chapters or prospectively for new monographs or chapters.
The three pharmacopeias have a commitment to respect the agreed working procedures and the associated time deadlines as an essential part of the harmonization procedure.
Harmonization of pharmacopeial documents in the PDG is performed on the basis of decisions of the expert bodies of each pharmacopeia. The PDG works transparently in many ways, but principally through the public notice and comment procedures of each pharmacopeia.
Where necessary, meetings of experts are held to identify potential solutions to difficult problems.
The specific stages of the PDG process involved in harmonization are described below.
Stage 1: Identification
On the basis of an inquiry among its users, the PDG identifies subjects to be harmonized among PDG pharmacopeias and nominates a coordinating pharmacopeia for each subject.
The PDG distributes the work by consensus among the three pharmacopeias and strives for a balance in the distribution of assignments to coordinating pharmacopeias.
Stage 2: Investigation
For a subject to be harmonized retrospectively, the coordinating pharmacopeia collects the information on the existing specifications in the three pharmacopeias, on the grades of products marketed, and on the potential analytical procedures.
The coordinating pharmacopeia prepares a draft monograph or chapter, accompanied by a report giving the rationale for the proposal with validation data.
Stage 2 ends with the proposal draft, which is mentioned in this procedure as a Stage 3 draft. The Stage 3 draft, accompanied by supporting comments or data that explain the reasons for each test procedure or limit proposed, is sent by the coordinating pharmacopeia to the secretariats of the other two PDG pharmacopeias.
Stage 3: Proposal for Expert Committee Review
The three pharmacopeias forward the Stage 3 draft to their expert committee (through meetings or consultation by correspondence).
Comments by the experts resulting from this preliminary survey are sent to their respective pharmacopeial secretariat, preferably within 2 months. However, the comment period should not exceed 4 months. Within 2 months of receipt of the comments, the pharmacopeial secretariat should consolidate the comments and forward them to the coordinating pharmacopeia.
The coordinating pharmacopeia reviews the comments received and prepares a harmonized document (Stage 4 draft) accompanied by a commentary discussing comments received about the previous text and providing reasons for action taken in response to those comments.
The Stage 4 draft, as far as possible written in global style—a style easily understood by a variety of readers—together with the commentary, are sent to the secretariats of the other pharmacopeias (end of Stage 3).
Stage 4: Official Inquiry
The Stage 4 draft and the commentary are published in the revision document of each pharmacopeia in a section entitled International Harmonization. The draft is published in its entirety.
The corresponding secretariats may have to add information essential to the understanding of the implementation of the texts (e.g., the description of an analytical procedure or of reagents that do not exist in the pharmacopeia), and a translation is added by the European and Japanese Pharmacopoeias. The style may be adapted to that of the pharmacopeia concerned, or global style may be used. A pharmacopeia can add text, either to amplify some of the requirements with additional information or because national requirements and compendial policy dictate that the addition is necessary. However, there must be a clear indication that this additional information is not part of the harmonized document. This will avoid additional text being included after the harmonization process is completed, but will allow interested parties to review a complete text. The three pharmacopeias endeavor to publish the drafts simultaneously or as close together as possible.
Comments regarding this draft are sent by readers of the revision document to their respective pharmacopeial secretariat, preferably within 4 months and at most within 6 months of its publication.
Each pharmacopeia analyzes the comments received and submits its consolidated comments to the coordinating pharmacopeia within 2 months of the end of the review or comment period.
The coordinating pharmacopeia reviews the comments received and prepares a draft harmonized document (Stage 5A draft), accompanied by a commentary discussing comments received regarding the previous text and providing reasons for action taken in response to those comments.
The Stage 5A draft, together with the commentary, is sent to the secretariats of the other two PDG pharmacopeias.
Stage 5. Consensus
5A. PROVISIONAL
The Stage 5A draft is reviewed and commented on by the other two PDG pharmacopeias within 4 months of receipt. The three pharmacopeias shall do their utmost to reach full agreement at this stage to obtain a final consensus document.
If a consensus has not been reached, the coordinating pharmacopeia prepares a revised version (Stage 5A/2), taking into consideration relevant, substantiated comments on the Stage 5A document from the two other pharmacopeias. The revised document (Stage 5A/2), together with the commentary, is sent to the secretariats of the other two PDG pharmacopeias. The revised document is reviewed and commented on by the other two PDG pharmacopeias, preferably within 2 months of receipt. This review or comment and revision process of the 5A document is repeated (Stage 5A/n) until the three PDG pharmacopeias reach a consensus or until the coordinating pharmacopeia considers that harmonization by attribute should be applied.
If the coordinating pharmacopeia considers certain attributes in the monograph or provisions in a general chapter (especially for retroactive harmonization) are such that it will not be possible to harmonize within a reasonable time period, harmonization by attribute will be applied. If harmonization by attribute is applied, a special cover page (see the table in the Appendix) indicating harmonization is included with the draft. The text contains harmonized attributes and provisions, and nonharmonized and local attributes are not included.The nonharmonized attributes are clearly indicated in the text as such. The table is prepared as follows: if three pharmacopeias agree on the attribute, there will be a (+) in all columns; if two pharmacopeias agree that the attribute should be included and have agreed on the method and limit, there will be a (+) in the column for those two pharmacopeias, and a (–) in the column for the pharmacopeia that will not stipulate the test.
For nonharmonized or local requirements, if three pharmacopeias agree that the attribute should be included, but have not come to agreement on the method or limit: state attribute under “nonharmonized attributes.” If only one pharmacopeia will include an attribute: state under “local requirement.”
If the Stage 5A draft is substantially different from the Stage 4 draft, the PDG may decide that it should be published again in the revision documents; the draft then reverts technically to Stage 4, revised.
5B. DRAFT SIGN-OFF
When agreement is reached, the 5B draft is sent by the coordinating pharmacopeia to the other pharmacopeias no later than 4 weeks before a PDG meeting for final confirmation. The document is then presented for sign-off at the PDG meeting.This document includes nonharmonized attributes clearly marked as such.
Stage 6: Regional Adoption and Implementation
NOTE—The last two stages of the implementation of the “harmonized” chapters and monographs take place independently according to the procedures established by each pharmacopeial organization.
6A. Regional Adoption
The document is submitted for adoption to the organization responsible for each pharmacopeia. Each pharmacopeia incorporates the harmonized draft according to its own procedures. Stylistic and editorial differences may occur.
Adopted texts are published by the three pharmacopeias in their supplements, or where applicable, in a new edition.
If necessary, the Stage 5B draft may be adopted with some amendments (local requirements) corresponding to a general policy in the national or regional (European) area. If a pharmacopeia includes a local attribute after the sign-off of a text, it will inform the PDG. It is, however, preferred to include the nonharmonized text in Stage 5B as an alert to the other pharmacopeias that there will be some differences in text in the final document.
Users of the pharmacopeias are appropriately informed of the harmonization status of monographs and general chapters. In the European Pharmacopoeia (EP) and USP–NF, for general chapters, this is done via a preliminary paragraph. For the Japanese Pharmacopoeia (JP), a notification is made by the MHLW, and information is given in a general chapter.
6B. Implementation
The pharmacopeias will inform each other of the date of implementation in their particular region.
The date of implementation of a harmonized document varies in the three PDG regions depending on their legal requirements, need of translation, and publication schedules. Each pharmacopeia generally allows some period of time after publication for implementation to allow manufacturers and other users to achieve conformity. Harmonization is not achieved until the text becomes official in all three pharmacopeias.
Stage 7: Inter-Regional Implementation
When a harmonized text has become official in all three pharmacopeias, EP and USP publish a statement indicating the harmonization status of the text; JP publishes a statement to the same effect at Stage 6B. These statements are intended to promote regulatory acceptance of interchangeability of harmonized monographs and general chapters.
Revision
The procedure for the revision of harmonized monographs and chapters is as follows.
The pharmacopeias participating in the PDG have agreed not to revise unilaterally any harmonized document (monograph or chapter) after sign-off or after publication.
A pharmacopeia requesting the revision of a monograph or chapter shall apply the following criteria for justification of the revision:
-
Public health and safety reasons.
-
Insufficient supply of pharmacopeial-quality product on the market.
-
Specified analytical reagents or equipment unavailable.
-
New methods of preparation of products or reagents not covered by the current monograph.
-
Analytical procedures capable of being replaced by more appropriate, accurate, or precise procedures.
The PDG as a whole has to agree to initiate the revision. A coordinating pharmacopeia will be nominated. The coordinating pharmacopeia will prepare a Stage 3 draft, based on the validation of data provided by the pharmacopeia requesting the revision.
The PDG Working Procedures will then be followed. The revisions of a sign-off document prepared for this or other reasons are indicated as revision 1, 2, 3, etc.
In case of health and safety issues, and whenever agreed to by the PDG, an accelerated procedure shall be applied (shortening or eliminating stages).
Discussion
Harmonization of general chapters and monographs benefits manufacturers of pharmaceutical products intended for human use, regulatory agencies, and ultimately, practitioners and patients. Benefits are derived from (1) reduced development effort; (2) simplification of regulatory filings; and (3) reduced release testing.
Pharmacopeial harmonization amplifies the work of the ICH, particularly for Quality topics. While the PDG is not part of the ICH, the PDG periodically provides updates to the ICH Steering Committee, and in the past participated in a joint task force. This task force focused on harmonization of general chapters considered important to the ICH harmonized document Specifications: Test Procedures and Acceptance Criteria for New Drug Substances and New Drug Products: Chemical Substances (Q6A). USP also participates in the International Cooperation on Harmonization of Technical Requirements for Registration of Veterinary Products (VICH). As with the ICH, some of the quality guidelines developed in VICH depend upon harmonization of pharmacopeial general chapters. A major difference between the PDG and ICH/VICHs is that the ICH/VICH guidelines generally are applicable only to ingredients and drug products not previously registered in an ICH/VICH region or nation, whereas the PDG harmonization applies to all marketed products in the applicable region or nation.
In the case of harmonization by attribute, nonharmonization or divergence will be indicated in
USP–NF and
EP by the symbol
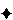
. For these nonharmonized attributes, reliance upon the individual pharmacopeia is required. A monograph or general chapter in one PDG pharmacopeia may unilaterally include additional local or national attributes that are not included in the corresponding text of the other pharmacopeias. Such text is not considered by the PDG to be a divergence from the PDG harmonized text.
As with other
USP–NF revisions, draft harmonization texts are published for comment in
Pharmacopeial Forum. Final harmonized official text in
USP–NF is presented in the latest edition,
Supplement, or
Interim Revision Announcement. The current status of all harmonization projects appears in
Table 1 and
Table 2. These status tables will be updated in subsequent editions of
USP–NF and its
Supplements.
In the U.S., cases of noncompliance or dispute are resolved through performance of the official procedure in USP or NF. If the procedure and its acceptance criteria are harmonized in the PDG, then a manufacturer may follow the relevant compendial instructions in USP–NF, EP, or JP.
Table 1. Status of Harmonization—Excipient
Monographs
| Excipient Name |
Coordinating
Pharmacopeia |
Harmonization
Stage |
| Alcohol |
EP |
6 |
| Benzyl Alcohol |
EP |
6 |
| Dehydrated Alcohol |
EP |
6 |
| Butylparaben |
EP |
6 |
| Calcium Carbonate |
USP |
2 |
| Calcium Disodium Edetate |
JP |
5A2 |
Calcium Phosphate Dibasic
 (and anhydrous) (and anhydrous) |
JP |
5B |
Carboxymethylcellulose
 Calcium Calcium |
USP |
6 |
Carboxymethylcellulose
 Sodium Sodium |
USP |
4 |
| Carmellose |
JP |
2 |
| Cellulose Acetate |
USP |
6 |
| Cellulose Acetate Phthalate |
USP |
6 |
| Microcrystalline Cellulose |
USP |
6 |
| Cellulose, Powdered |
USP |
6 |
| Citric Acid, Anhydrous |
EP |
6 |
| Citric Acid, Monohydrate |
EP |
6 |
| Copovidone |
JP |
2 |
| Croscarmellulose Sodium |
USP |
6 |
| Crospovidone |
EP |
4 |
| Ethylcellulose |
EP |
6 |
| Ethylparaben |
EP |
6 |
| Gelatin |
EP |
2 |
| Glucose |
EP |
2 |
| Glycerin |
USP |
3 |
| Glyceryl Monostearate |
USP |
2 |
| Hydroxyethyl Cellulose |
EP |
4 |
| Hydroxypropyl Cellulose |
USP |
4 |
Hydroxypropyl Cellulose,
 Low Substituted Low Substituted |
USP |
4 |
Hydroxypropylmethyl
 Cellulose Cellulose |
JP |
6 |
Hydroxypropylmethyl
 Cellulose Phthalate Cellulose Phthalate |
USP |
5A |
| Lactose, Anhydrous |
USP |
6 |
| Lactose, Monohydrate |
USP |
6 |
| Magnesium Stearate |
USP |
4 |
| Mannitol |
EP |
2 |
| Methylcellulose |
JP |
6 |
| Methylparaben |
EP |
6 |
| Petrolatum, White |
USP |
4 |
| Polyethylene Glycol |
USP |
4 |
| Polysorbate 80 |
EP |
3 |
| Povidone |
JP |
5A |
| Propylene Glycol |
EP |
3 |
| Propylparaben |
EP |
6 |
| Saccharin |
USP |
6 |
| Saccharin, Calcium |
USP |
6 |
| Saccharin, Sodium |
USP |
6 |
| Silicon Dioxide |
JP |
4 |
| Silicon Dioxide, Colloidal |
JP |
4 |
| Sodium Chloride |
EP |
6 |
| Sodium Lauryl Sulfate |
USP |
2 |
| Sodium Starch Glycolate |
USP |
6 |
| Starch, Corn |
USP |
6 |
| Starch, Potato |
EP |
6 |
| Starch, Pregelatinized |
JP |
2 |
| Starch, Rice |
EP |
4 Revised |
| Starch, Wheat |
EP |
6 |
| Stearic Acid |
EP |
4 |
| Sucrose |
EP |
3 |
| Talc |
EP |
6 |
| Titanium Dioxide |
JP |
5A2 |
Table 2. Status of Harmonization—General Chapters
Harmonized Monographs and Chapters
HARMONIZED CHAPTERS
 85
85 Bacterial Endotoxins Test—
Bacterial Endotoxins Test—
This chapter has been harmonized by PDG and published in the
European Pharmacopoeia and in the
Japanese Pharmacopoeia. Portions of the chapter that are not harmonized with the other two pharmacopeias are marked by the symbol
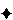
. Footnotes 1, 2, and 4 are in the
USP but are not in the
EP or
JP. These footnotes give additional information, such as the calculation of endotoxin limits for different classes of products (Footnote 4) or reference
USP chapters (Footnote 2).
The USP Endotoxin Reference Standard is harmonized with the International Reference Standard for Endotoxin and the EP Endotoxin Reference Standard and indirectly harmonized with the JP Endotoxin Reference Standard that is indexed to the International Reference Standard. The result is that 1 USP Endotoxin Unit = 1 International Endotoxin Unit = 1 EP Endotoxin Unit.
GLOSSARY
Harmonized monograph or general chapter—
Text that has reached Stage 5B and that has been countersigned by the three PDG pharmacopeias.
Total harmonization—
A monograph or a general chapter that is identical in the three PDG pharmacopeias in terms of identical tests, test procedures, and acceptance criteria.
Harmonization by attributes—
A monograph or a general chapter that contains a combination of harmonized and nonharmonized tests or sections.
Interchangeability—
A test or a section of a monograph or a general chapter that is not the same among the PDG pharmacopeias, but the accept/reject decision is the same regardless of which one of the tests or sections of the PDG pharmacopeia is used.
Local or national divergence—
A monograph or a general chapter in one of the PDG pharmacopeias contains specific attributes of local or national origin in addition to the PDG harmonized/nonharmonized attributes.
Nonharmonized attributes—
Attributes that could not be agreed upon by the three PDG pharmacopeias because of regional differences, regulatory differences, nonavailability of reagents, etc.