Importance of a Microbiological Evaluation Program for Controlled Environments
Monitoring of total particulate count in controlled environments, even with the use of electronic instrumentation on a continuous basis, does not provide information on the microbiological content of the environment. The basic limitation of particulate counters is that they measure particles of 0.5 µm or larger. While airborne microorganisms are not free-floating or single cells, they frequently associate with particles of 10 to 20 µm. Particulate counts as well as microbial counts within controlled environments vary with the sampling location and the activities being conducted during sampling. Monitoring the environment for nonviable particulates and microorganisms is an important control function because they both are important in achieving product compendial requirements for
Particulate Matter and
Sterility under
Injections  1
1
.
Microbial monitoring programs for controlled environments should assess the effectiveness of cleaning and sanitization practices by and of personnel that could have an impact on the bioburden of the controlled environment. Microbial monitoring, regardless of how sophisticated the system may be, will not and need not identify and quantitate all microbial contaminants present in these controlled environments. However, routine microbial monitoring should provide sufficient information to ascertain that the controlled environment is operating within an adequate state of control.
Environmental microbial monitoring and analysis of data by qualified personnel will permit the status of control to be maintained in clean rooms and other controlled environments. The environment should be sampled during normal operations to allow for the collection of meaningful data. Microbial sampling should occur when materials are in the area, processing activities are ongoing, and a full complement of operating personnel is on site.
Microbial monitoring of clean rooms and some other controlled environments, when appropriate, should include quantitation of the microbial content of room air, compressor air that enters the critical area, surfaces, equipment, sanitization containers, floors, walls, and personnel garments (e.g., gowns and gloves). The objective of the microbial monitoring program is to obtain representative estimates of bioburden of the environment. When data are compiled and analyzed, any trends should be evaluated by trained personnel. While it is important to review environmental results on the basis of recommended and specified frequency, it is also critical to review results over extended periods to determine whether trends are present. Trends can be visualized through the construction of statistical control charts that include alert and action levels. The microbial control of controlled environments can be assessed, in part, on the basis of these trend data. Periodic reports or summaries should be issued to alert the responsible manager.
When the specified microbial level of a controlled environment is exceeded, a documentation review and investigation should occur. There may be differences in the details of the investigation, depending on the type and processing of the product manufactured in the room. Investigation should include a review of area maintenance documentation; sanitization documentation; the inherent physical or operational parameters, such as changes in environmental temperature and relative humidity; and the training status of personnel involved. Following the investigation, actions taken may include reinforcement of training of personnel to emphasize the microbial control of the environment; additional sampling at increased frequency; additional sanitization; additional product testing; identification of the microbial contaminant and its possible source; and an evaluation of the need to reassess the current standard operating procedures and to revalidate them, if necessary.
Based on the review of the investigation and testing results, the significance of the microbial level being exceeded and the acceptability of the operations or products processed under that condition may be ascertained. Any investigation and the rationale for the course of action should be documented and included as part of the overall quality management system.
A controlled environment such as a clean zone or clean room is defined by certification according to a relevant clean room operational standard. Parameters that are evaluated include filter integrity, air velocity, air patterns, air changes, and pressure differentials. These parameters can affect the microbiological bioburden of the clean room operation. The design, construction, and operation of clean rooms varies greatly, making it difficult to generalize requirements for these parameters. An example of a method for conducting a particulate challenge test to the system by increasing the ambient particle concentration in the vicinity of critical work areas and equipment has been developed by Ljungquist and Reinmuller.
1 First, smoke generation allows the air movements to be visualized throughout a clean room or a controlled environment. The presence of vortices or turbulent zones can be visualized, and the airflow pattern may be fine-tuned to eliminate or minimize undesirable effects. Then, particulate matter is generated close to the critical zone and
sterile field. This evaluation is done under simulated production conditions, but with equipment and personnel in place.
Proper testing and optimization of the physical characteristics of the clean room or controlled environment is essential prior to completion of the validation of the microbiological monitoring program. Assurance that the controlled environment is operating adequately and according to its engineering specifications will give a higher assurance that the bioburden of the environment will be appropriate for aseptic processing. These tests should be repeated during routine certification of the clean room or controlled environment and whenever changes made to the operation, such as personnel flow, processing, operation, material flow, air-handling systems, or equipment layout, are determined to be significant.
Training of Personnel
Aseptically processed products require manufacturers to pay close attention to detail and to maintain rigorous discipline and strict supervision of personnel in order to maintain the level of environmental quality appropriate for the sterility assurance of the final product.
Training of all personnel working in controlled environments is critical. This training is equally important for personnel responsible for the microbial monitoring program, where contamination of the clean working area could inadvertently occur during microbial sampling. In highly automated operations, the monitoring personnel may be the employees who have the most direct contact with the critical zones within the processing area. Monitoring of personnel should be conducted before or after working in the processing area.
Microbiological sampling has the potential to contribute to microbial contamination due to inappropriate sampling techniques. A formal personnel training program is required to minimize this risk. This formal training should be documented for all personnel entering controlled environments.
Management of the facility must assure that all personnel involved in operations in clean rooms and controlled environments are well versed in relevant microbiological principles. The training should include instruction on the basic principles of aseptic processing and the relationship of manufacturing and handling procedures to potential sources of product contamination. This training should include instruction on the basic principles of microbiology, microbial physiology, disinfection and sanitation, media selection and preparation, taxonomy, and sterilization as required by the nature of personnel involvement in aseptic processing. Personnel involved in microbial identification will require specialized training on required laboratory methods. Additional training on the management of the environmental data collected must be provided to personnel. Knowledge and understanding of applicable standard operating procedures is critical, especially those standard operating procedures relating to corrective measures that are taken when environmental conditions so dictate. Understanding of regulatory compliance policies and each individual's responsibilities with respect to good manufacturing practices (GMPs) should be an integral part of the training program as well as training in conducting investigations and in analyzing data.
The major source of microbial contamination of controlled environments is the personnel. Contamination can occur from the spreading of microorganisms by individuals, particularly those with active infections. Only healthy individuals should be permitted access to controlled environments.
These facts underscore the importance of good personal hygiene and a careful attention to detail in the aseptic gowning procedure used by personnel entering the controlled environment. Once these employees are properly gowned—including complete facial coverage—they must be careful to maintain the integrity of their gloves and suits at all times. Since the major threat of contamination of product being aseptically processed comes from the operating personnel, the control of microbial contamination associated with these personnel is one of the most important elements of the environmental control program.
The importance of thorough training of personnel working in controlled environments, including aseptic techniques, cannot be overemphasized. The environmental monitoring program, by itself, will not be able to detect all events in aseptic processing that could compromise the microbiological quality of the environment. Therefore, periodic media-fill or process simulation studies to revalidate the process are necessary to assure that the appropriate operating controls and training are effectively maintained.
Critical Factors Involved in the Design and Implementation of a Microbiological Environmental Control Program
An environmental control program should be capable of detecting an adverse drift in microbiological conditions in a timely manner that would allow for meaningful and effective corrective actions. It is the responsibility of the manufacturer to develop, initiate, implement, and document such a microbial environmental monitoring program.
Although general recommendations for an environmental control program will be discussed, it is imperative that such a program be tailored to specific facilities and conditions. A general microbiological growth medium such as Soybean Casein Digest Medium should be suitable in most cases. This medium may be supplemented with additives to overcome or to minimize the effects of sanitizing agents, or of antibiotics if used or processed in these environments. The detection and quantitation of yeasts and molds should be considered. General mycological media, such as Sabouraud's, Modified Sabouraud's, or Inhibitory Mold Agar are acceptable. Other media that have been validated for promoting the growth of fungi, such as Soybean–Casein Digest Agar, can be used. In general, testing for obligatory anaerobes is not performed routinely. However, should conditions or investigations warrant, such as the identification of these organisms in sterility testing facilities, more frequent testing is indicated. The ability of the selected media to detect and quantitate these anaerobes or microaerophilic microorganisms should be evaluated.
The selection of time and incubation temperatures is made once the appropriate media have been selected. Typically, incubation temperatures in the 22.5 ± 2.5

and 32.5 ± 2.5

ranges have been used with an incubation time of 72 and 48 hours, respectively. Sterilization processes used to prepare growth media for the environmental program should be validated and, in addition, media should be examined for sterility and for growth promotion as indicated under
Sterility Tests  71
71
. In addition, for the
Growth Promotion test, representative microflora isolated from the controlled environment or ATCC strain preparations of these isolates may also be used to test media. Media must be able to support growth when inoculated with less than 100 colony-forming units (cfu) of the challenge organisms.
An appropriate environmental control program should include identification and evaluation of sampling sites and validation of methods for microbiological sampling of the environment.
The methods used for identification of isolates should be verified using indicator microorganisms (see
Microbial Limit Tests  61
61
).
Establishment of Microbiological Alert and Action Levels in Controlled Environments
The principles and concepts of statistical process control are useful in establishing Alert and Action levels and in reacting to trends.
An Alert level in microbiological environmental monitoring is that level of microorganisms that shows a potential drift from normal operating conditions. Exceeding the Alert level is not necessarily grounds for definitive corrective action, but it should at least prompt a documented follow-up investigation that could include sampling plan modifications.
An Action level in microbiological environmental monitoring is that level of microorganisms that when exceeded requires immediate follow-up and, if necessary, corrective action.
Alert levels are usually based upon historical information gained from the routine operation of the process in a specific controlled environment.
In a new facility, these levels are generally based on prior experience from similar facilities and processes; and at least several weeks of data on microbial environmental levels should be evaluated to establish a baseline.
These levels are usually re-examined for appropriateness at an established frequency. When the historical data demonstrate improved conditions, these levels can be re-examined and changed to reflect the conditions. Trends that show a deterioration of the environmental quality require attention in determining the assignable cause and in instituting a corrective action plan to bring the conditions back to the expected ranges. However, an investigation should be implemented and an evaluation of the potential impact this has on a product should be made.
Methodology and Instrumentation for Quantitation of Viable Airborne Microorganisms
It is generally accepted by scientists that airborne microorganisms in controlled environments can influence the microbiological quality of the intermediate or final products manufactured in these areas. Also, it generally is accepted that estimation of the airborne microorganisms can be affected by instruments and procedures used to perform these assays. Therefore, where alternative methods or equipment is used, the general equivalence of the results obtained should be ascertained. Advances in technology in the future are expected to bring innovations that would offer greater precision and sensitivity than the current available methodology and may justify a change in the absolute numbers of organisms that are detected.
Today, the most commonly used samplers in the U.S. pharmaceutical and medical device industry are the impaction and centrifugal samplers. A number of commercially available samplers are listed for informational purposes. The selection, appropriateness, and adequacy of using any particular sampler is the responsibility of the user.
Slit-to-Agar Air Sampler (STA)—
This sampler is the instrument upon which the microbial guidelines given in
Table 3 for the various controlled environments are based. The unit is powered by an attached source of controllable vacuum. The air intake is obtained through a standardized slit below which is placed a slowly revolving Petri dish containing a nutrient agar. Particles in the air that have sufficient mass impact on the agar surface and viable organisms are allowed to grow out. A remote air intake is often used to minimize disturbance of the laminar flow field.
Sieve Impactor—
The apparatus consists of a container designed to accommodate a Petri dish containing a nutrient agar. The cover of the unit is perforated, with the perforations of a predetermined size. A vacuum pump draws a known volume of air through the cover, and the particles in the air containing microorganisms impact on the agar medium in the Petri dish. Some samplers are available with a cascaded series of containers containing perforations of decreasing size. These units allow for the determination of the distribution of the size ranges of particulates containing viable microorganisms, based on which size perforations admit the particles onto the agar plates.
Centrifugal Sampler—
The unit consists of a propeller or turbine that pulls a known volume of air into the unit and then propels the air outward to impact on a tangentially placed nutrient agar strip set on a flexible plastic base.
Sterilizable Microbiological Atrium—
The unit is a variant of the single-stage sieve impactor. The unit's cover contains uniformly spaced orifices approximately 0.25 inch in size. The base of the unit accommodates one Petri dish containing a nutrient agar. A vacuum pump controls the movement of air through the unit, and a multiple-unit control center as well as a remote sampling probe are available.
Surface Air System Sampler—
This integrated unit consists of an entry section that accommodates an agar contact plate. Immediately behind the contact plate is a motor and turbine that pulls air through the unit's perforated cover over the agar contact plate and beyond the motor, where it is exhausted. Multiple mounted assemblies are also available.
Gelatin Filter Sampler—
The unit consists of a vacuum pump with an extension hose terminating in a filter holder that can be located remotely in the critical space. The filter consists of random fibers of gelatin capable of retaining airborne microorganisms. After a specified exposure time, the filter is aseptically removed and dissolved in an appropriate diluent and then plated on an appropriate agar medium to estimate its microbial content.
Settling Plates—
This method is still widely used as a simple and inexpensive way to qualitatively assess the environments over prolonged exposure times. The exposure of open agar-filled Petri dishes, or settling plates, is not to be used for quantitative estimations of the microbial contamination levels of critical enviroments.
One of the major limitations of mechanical air samplers is the limitation in sample size of air being sampled. Where the microbial level in the air of a controlled environment is expected to contain not more than three cfu per cubic meter, several cubic meters of air should be tested if results are to be assigned a reasonable level of precision and accuracy. Often this is not practical. To show that microbial counts present in the environment are not increasing over time, it might be necessary to extend the time of sampling to determine if the time of sampling is a limiting factor or not. Typically, slit-to-agar samplers have an 80-liter-per-minute sampling capacity (the capacity of the surface air system is somewhat higher). If one cubic meter of air is tested, then it would require an exposure time of 15 minutes. It may be necessary to use sampling times in excess of 15 minutes to obtain a representative environmental sample. Although there are samplers reported to be capable of very high sampling volume rates, consideration in these situations should be given to the potential for disruption of the airflow patterns in any critical area or to the creation of a turbulence that could increase the probability of contamination.
For centrifugal air samplers, a number of earlier studies showed that the samples demonstrated a selectivity for larger particles. The use of this type of sampler may have resulted in higher airborne counts than the other types of air samplers because of that inherent selectivity.
When selecting a centrifugal sampler, the effect of the sampler on the linearity of the airflow in the controlled zone where it is placed for sampling should be taken into consideration. Regardless of the type of sampler used, the use of a remote probe requires determining that the extra tubing does not have an adverse effect on the viable airborne count. This effect should either be eliminated or, if this is not possible, a correction factor should be introduced in the reporting of results.
Operational Evaluation of the Microbiological Status of Aseptically Filled Products in Clean Rooms and Other Controlled Environments
The controlled environment is monitored through an appropriate environmental monitoring program. To assure that minimal bioburden is achieved, additional information on the evaluation of the microbiological status of the controlled environment can be obtained by the use of media fills. An acceptable media fill shows that a successful simulated product run can be conducted on the manufacturing line at that point in time. However, other factors are important, such as appropriate construction of facilities, environmental monitoring and training of personnel.
When an aseptic process is developed and installed, it is generally necessary to qualify the microbiological status of the process by running at least three successful consecutive media fills. A media fill utilizes growth medium in lieu of products to detect the growth of microorganisms. Issues in the development of a media fill program that should be considered are the following: media-fill procedures, media selection, fill volume, incubation, time and temperature, inspection of filled units, documentation, interpretation of results, and possible corrective actions required.
Since a media fill is designed to simulate aseptic processing of a specified product, it is important that conditions during a normal product run are in effect during the media fill. This includes the full complement of personnel and all the processing steps and materials that constitute a normal production run. During the conduct of media fill, various predocumented interventions that are known to occur during actual product runs should be planned (e.g., changing filling needles, fixing component jams).
Alternatively, in order to add a safety margin, a combination of possible conditions can be used. Examples may include frequent start and stop sequences, unexpected repair of processing system, replacement of filters, etc. The qualification of an aseptic process need not be done for every product, but should be done for each processing line. Since the geometry of the container (size as well as opening of the container) and the speed of the line are factors that are variable in the use of an aseptic processing line, appropriate combination of these factors, preferably at the extremes, should be used in the qualification of the line. A rationale for products used should be documented.
The 1987 FDA Guideline on Sterile Drug Products Produced by Aseptic Processing indicates that media-fill runs be done to cover all production shifts for line/product/container combinations. This guideline should be considered not only for qualification media-fill runs, but also for periodic reevaluation or revalidation. Media fill programs should also simulate production practices over extended runs. This can be accomplished by doing media-fill runs at the end of production runs.
In general, an all-purpose, rich medium such as Soybean Casein Broth that has been checked for growth promotion with a battery of indicator organisms (see
Sterility Tests  71
71
) at a level of below 100 cfu/unit, can be used. Isolates from the controlled environment where aseptic processing is to be conducted may also be used. Following the aseptic processing of the medium, the filled containers are incubated at 22.5 ± 2.5

or at 32.5 ± 2.5

. All media filled containers should be incubated for a minimum of 14 days. If two temperatures are used for incubation of media filled samples, then these filled containers should be incubated for at least 7 days at each temperature. Following incubation, the medium-filled containers should be inspected for growth. Media filled isolates are identified by genus and, when possible, by species in order to investigate the sources of contamination.
Critical issues in performing media fills are the number of fills to qualify an aseptic process, the number of units filled per media fill, the interpretation of results, and implementation of corrective actions. Historically, three media-fill runs during initial qualification or start-up of a facility are conducted to demonstrate consistency of the aseptic processing line. The minimum number of units to demonstrate a contamination rate of not more than 0.1%, which is the criterion for acceptance of a successful media-fill run, is at least 3,000. It should be emphasized that many firms in the United States and other countries are filling more than 3,000 units in a single media-fill run.
4 Pilot plant facilities used for preparing small clinical lots may use smaller media fills.
A number of international documents (i.e., ISO and EU-GMP) have also cited an expectation of zero positives out of 3,000 media filled units at the 95% confidence level. However, it is recognized that repeated media runs are required in order to confirm the statistical validity of the observed contamination rate for the process.
PDA Technical Monograph Number 17,
4 “A Survey of Current Sterile Manufacturing Practices,” indicated that many manufacturers believe that their aseptic processes are capable of contamination rates below 0.1%.
Since the most critical source of contamination in the clean room is the personnel, visual documentation that can be helpful in correlating production activities to contamination events during media fills is encouraged. The widespread use of isolator systems for sterility testing has demonstrated that elimination of personnel does reduce contamination in aseptic handling.
An Overview of the Emerging Technologies for Advanced Aseptic Processing
Because of the strong correlation between human involvement and intervention and the potential for product contamination in aseptic processing, production systems in which personnel are removed from critical zones have been designed and implemented. Methods developed to reduce the likelihood of contamination include equipment automation, barriers, and isolator systems. Facilities that employ these advanced aseptic processing strategies are already in operation. In facilities where personnel have been completely excluded from the critical zone, the necessity for room classification based on particulate and environmental microbiological monitoring requirements may be significantly reduced.
The following are definitions of some of the systems currently in place to reduce the contamination rate in aseptic processing:
Barriers—
In the context of aseptic processing systems, a barrier is a device that restricts contact between operators and the aseptic field enclosed within the barrier. These systems are used in hospital pharmacies, laboratories, and animal care facilities, as well as in aseptic filling. Barriers may not be sterilized and do not always have transfer systems that allow passage of materials into or out of the system without exposure to the surrounding environment. Barriers range from plastic curtains around the critical production zones to rigid enclosures found on modern aseptic-filling equipment. Barriers may also incorporate such elements as glove ports, half-suits, and rapid-transfer ports.
Blow/Fill/Seal—
This type of system combines the blow-molding of container with the filling of product and a sealing operation in one piece of equipment. From a microbiological point of view, the sequence of forming the container, filling with sterile product, and formation and application of the seal are achieved aseptically in an uninterrupted operation with minimal exposure to the environment. These systems have been in existence for about 30 years and have demonstrated the capability of achieving contamination rates below 0.1%. Contamination rates of 0.001% have been cited for blow/fill/seal systems when combined media-fill data are summarized and analyzed.
Isolator—
This technology is used for a dual purpose. One is to protect the product from contamination from the environment, including personnel, during filling and closing, and the other is to protect personnel from deleterious or toxic products that are being manufactured.
Isolator technology is based on the principle of placing previously sterilized components (containers/products/closures) into a sterile environment. These components remain sterile during the whole processing operation, since no personnel or nonsterile components are brought into the isolator. The isolator barrier is an absolute barrier that does not allow for interchanges between the protected and unprotected environments. Isolators either may be physically sealed against the entry of external contamination or may be effectively sealed by the application of continuous overpressure. Manipulations of materials by personnel are done via use of gloves, half-suits, or full suits. All air entering the isolator passes through either an HEPA or UPLA filter, and exhaust air typically exits through an HEPA-grade filter. Peracetic acid and hydrogen peroxide vapor are commonly used for the surface sterilization of the isolator unit's internal environment. The sterilization of the interior of isolators and all contents are usually validated to a sterility assurance level of 10
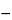 6
6.
Equipment, components, and materials are introduced into the isolator through a number of different procedures: use of a double-door autoclave; continuous introduction of components via a conveyor belt passing through a sterilizing tunnel; use of a transfer container system through a docking system in the isolator enclosure. It is also necessary to monitor closely an isolator unit's integrity, calibration, and maintenance.
The requirements for controlled environments surrounding these newer technologies for aseptic processing depend on the type of technology used.
Blow/Fill/Seal equipment that restricts employee contact with the product may be placed in a controlled environment, especially if some form of employee intervention is possible during production.
Barrier systems will require some form of controlled environment. Because of the numerous barrier system types and applications, the requirements for the environment surrounding the barrier system will vary. The design and operating strategies for the environment around these systems will have to be developed by the manufacturers in a logical and rational fashion. Regardless of these strategies, the capability of the system to produce sterile products must be validated to operate in accordance with pre-established criteria.
In isolators, the air enters the isolator through integral filters of HEPA quality or better, and their interiors are sterilized typically to a sterility assurance level of 10
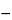 6
6; therefore, isolators contain sterile air, do not exchange air with the surrounding environment, and are free of human operators. However, it has been suggested that when the isolator is in a controlled environment, the potential for contaminated product is reduced in the event of a pinhole leak in the suit or glove.
The extent and scope of an environmental microbiological monitoring of these advanced systems for aseptic processing depends on the type of system used. Manufacturers should balance the frequency of environmental sampling systems that require human intervention with the benefit accrued by the results of that monitoring. Since barrier systems are designed to reduce human intervention to a minimum, remote sampling systems should be used in lieu of personnel intervention. In general, once the validation establishes the effectiveness of the barrier system, the frequency of sampling to monitor the microbiological status of the aseptic processing area could be reduced, as compared to the frequency of sampling of classical aseptic processing systems.
Isolator systems require relatively infrequent microbiological monitoring. Continuous total particulate monitoring can provide assurance that the air filtration system within the isolator is working properly. The methods for quantitative microbiological air sampling described in this chapter may not have sufficient sensitivity to test the environment inside an isolator. Experience with isolators indicates that under normal operations pinhole leaks or tears in gloves represent the major potential for microbiological contamination; therefore, frequent testing of the gloves for integrity and surface monitoring of the gloves is essential. Surface monitoring within the isolator may also be beneficial on an infrequent basis.
GLOSSARY
Airborne Particulate Count (also referred to as Total Particulate Count)—Particles detected are 0.5 µm and larger. When a number of particles is specified, it is the maximum allowable number of particles per cubic meter of air (or per cubic foot of air).
Airborne Viable Particulate Count (also referred to as Total Airborne Aerobic Microbial Count)—When a number of microorganisms is specified, it is the maximum number of colony-forming units (cfu) per cubic meter of air (or per cubic foot of air) that is associated with a Cleanliness Class of controlled environment based on the Airborne Particulate Count.
Aseptic Processing—A mode of processing pharmaceutical and medical products that involves the separate sterilization of the product and of the package (containers/closures or packaging material for medical devices) and the transfer of the product into the container and its closure under microbiologic critically controlled conditions.
Air Sampler—Devices or equipment used to sample a measured amount of air in a specified time to quantitate the particulate or microbiological status of air in the controlled environment.
Air Changes—The frequency per unit of time (minutes, hours, etc.) that the air within a controlled environment is replaced. The air can be recirculated partially or totally replaced.
Action Levels—Microbiological levels in the controlled environment, specified in the standard operating procedures, which when exceeded should trigger an investigation and a corrective action based on the investigation.
Alert Levels—Microbial levels, specified in the standard operating procedures, which when exceeded should result in an investigation to ensure that the process is still within control. Alert levels are specific for a given facility and are established on the basis of a baseline developed under an environmental monitoring program. These Alert levels can be modified depending on the trend analysis done in the monitoring program. Alert levels are always lower than Action levels.
Bioburden—Total number of microorganisms detected in or on an article.
Clean Room—A room in which the concentration of airborne particles is controlled to meet a specified airborne particulate Cleanliness Class. In addition, the concentration of microorganisms in the environment is monitored; each Cleanliness Class defined is also assigned a microbial level for air, surface, and personnel gear.
Clean Zone—A defined space in which the concentration of airborne particles and microorganisms are controlled to meet specific Cleanliness Class levels.
Controlled Environment—Any area in an aseptic process system for which airborne particulate and microorganism levels are controlled to specific levels, appropriate to the activities conducted within that environment.
Commissioning of a Controlled Environment—Certification by engineering and quality control that the environment has been built according to the specifications of the desired cleanliness class and that, under conditions likely to be encountered under normal operating conditions (or worst-case conditions), it is capable of delivering an aseptic process. Commissioning includes media-fill runs and results of the environmental monitoring program.
Corrective Action—Actions to be performed that are in standard operating procedures and that are triggered when certain conditions are exceeded.
Environmental Isolates—Microorganisms that have been isolated from the environmental monitoring program.
Environmental Monitoring Program—Documented program, implemented through standard operating procedures, that describes in detail the procedures and methods used for monitoring particulates as well as microorganisms in controlled environments (air, surface, personnel gear). The program includes sampling sites, frequency of sampling, and investigative and corrective actions that should be followed if Alert or Action levels are exceeded. The methodology used for trend analysis is also described.
Equipment Layout—Graphical representation of an aseptic processing system that denotes the relationship between and among equipment and personnel. This layout is used in the Risk Assessment Analysis to determine sampling site and frequency of sampling based on potential for microbiological contamination of the product/container/closure system. Changes must be assessed by responsible managers, since unauthorized changes in the layout for equipment or personnel stations could result in increase in the potential for contamination of the product/container/closure system.
Federal Standard 209E—“Airborne Particulate Cleanliness Classes in Clean Rooms and Clean Zones” is a standard approved by the Commissioner, Federal Supply Services, General Service Administration, for the use of “All Federal Agencies.” The Standard establishes classes of air cleanliness based on specified concentration of airborne particulates. These classes of air cleanliness have been developed, in general, for the electronic industry “super-clean” controlled environments. In the pharmaceutical industry, the Federal Standard 209E is used to specify the construction of controlled environment. Class 100, Class 10,000, and Class 100,000 are generally represented in an aseptic processing system. If the classification system is applied on the basis of particles equal to or greater than 0.5 µm, these classes are now represented in the SI system by Class M3.5, M5.5, and M6.5, respectively.
Filter Integrity—Testing that ensures that a filter functional performance is satisfactory [e.g., dioctyl phthalate (DOP) and bubble point test].
Material Flow—The flow of material and personnel entering controlled environments should follow a specified and documented pathway that has been chosen to reduce or minimize the potential for microbial contamination of the product/closure/container systems. Deviation from the prescribed flow could result in increase in potential for microbial contamination. Material/personnel flow can be changed, but the consequences of the changes from a microbiological point of view should be assessed by responsible managers and must be authorized and documented.
Media Growth Promotion—Procedure that references
Growth Promotion under
Sterility Tests  71
71
to demonstrate that media used in the microbiological environmental monitoring program, or in
media-fill runs, are capable of supporting growth of indicator microorganisms and of environmental isolates from samples obtained through the monitoring program or their corresponding ATCC strains.
Media Fill—Microbiological simulation of an aseptic process by the use of growth media processed in a manner similar to the processing of the product and with the same container/closure system being used.
Out-of-Specification Event—Temporary or continuous event when one or more of the requirements included in standard operating procedures for controlled environments are not fulfilled.
Product Contact Areas—Areas and surfaces in a controlled environment that are in direct contact with either products, containers, or closures and the microbiological status of which can result in potential microbial contamination of the product/container/closure system. Once identified, these areas should be tested more frequently than non-product-contact areas or surfaces.
Risk Assessment Analysis—Analysis of the identification of contamination potentials in controlled environments that establish priorities in terms of severity and frequency and that will develop methods and procedures that will eliminate, reduce, minimize, or mitigate their potential for microbial contamination of the product/container/closure system.
Sampling Plan—A documented plan that describes the procedures and methods for sampling a controlled environment; identifies the sampling sites, the sampling frequency, and number of samples; and describes the method of analysis and how to interpret the results.
Sampling Sites—Documented geographical location, within a controlled environment, where sampling for microbiological evaluation is taken. In general, sampling sites are selected because of their potential for product/container/closure contacts.
Standard Operating Procedures—Written procedures describing operations, testing, sampling, interpretation of results, and corrective actions that relate to the operations that are taking place in a controlled environment and auxiliary environments. Deviations from standard operating procedures should be noted and approved by responsible managers.
Sterile Field—In aseptic processing or in other controlled environments, it is the space at the level of or above open product containers, closures, or product itself, where the potential for microbial contamination is highest.
Sterility—Within the strictest definition of sterility, an article is deemed sterile when there is complete absence of viable microorganisms. Absolute sterility cannot be practically demonstrated without testing every article in a batch. Sterility is defined in probabilistic terms, where the likelihood of a contaminated article is acceptably remote.
Swabs—Devices provided that are used to sample irregular as well as regular surfaces for determination of microbial status. The swab, generally composed of a stick with an absorbent extremity, is moistened before sampling and used to sample a specified unit area of a surface. The swab is then rinsed in sterile saline or other suitable menstruum and the contents plated on nutrient agar plates to obtain an estimate of the viable microbial load on that surface.
Trend Analysis—Data from a routine microbial environmental monitoring program that can be related to time, shift, facility, etc. This information is periodically evaluated to establish the status or pattern of that program to ascertain whether it is under adequate control. A trend analysis is used to facilitate decision-making for requalification of a controlled environment or for maintenance and sanitization schedules.