Vancomycin, monohydrochloride.
Vancomycin monohydrochloride.
(
Sa)-(3
S,6
R,7
R,22
R,23
S,26
S,36
R,38a
R)-44-[[2-
O-(3-Amino- 2,3,6-trideoxy-3-
C-methyl-
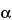
-
L-
lyxo-hexopyranosyl)-
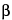
-
D-glucopyranosyl]oxy]-3-(carbamoylmethyl)-10,19-dichloro-2,3,4,5,6,7,23,24,25,26,36,37,38,38a-tetradecahydro- 7,22,28,30,32-pentahydroxy-6-[(2
R)-4-methyl-2-(methylamino)]valeramido]-2,5,24,38,39-pentaoxo-22
H-8,11:18,21-dietheno-23,36-(iminomethano)-13,16:31,35-dimetheno-1
H,16
H-[1,6,9]oxadiazacyclohexadecino[4,5-
m][10,2,16]benzoxadiazacyclotetracosine-26-carboxylic acid, monohydrochloride.
[3
S-[3
R*,6
S*(
S*),7
S*,22
S*,23
R*,26
R*,36
S*,38a
S*]]-3-(2-Amino-2-oxoethyl)-44-[[2-
O-(3-amino-2,3,6-trideoxy-3-
C-methyl-
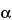
-
L-
lyxo-hexopyranosyl)-
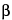
-
D-glucopyranosyl]oxy]-10,19-dichloro-2,3,4,5,6,7,23,24,25,26,36,37,38,38a-tetradecahydro-7,22,28,30,32-pentahydroxy-6-[[4-methyl-2-(methylamino)-1-oxopentyl]amino]-2,5,24,38,39-pentaoxo-22
H-8,11:18,21-dietheno-23,36-(iminomethano)-13,16:31,35-dimetheno-1
H,16
H-[1,6,9]oxadiazacyclohexadecino[4,5-
m][10,2,16]-benzoxadiazacyclotetracosine-26-carboxylic acid, monohydrochloride



[
1404-93-9].