Assay—
Accurately weigh about 1 g of Sulisobenzone, and dissolve in 20 mL of water in a conical flask. Add 200 mL of dehydrated isopropyl alcohol, titrate a portion with 0.1
N tetrabutylammonium hydroxide VS, and determine the two endpoints potentiometrically. Calculate the amount, in mg, of C
14H
12O
6S in the portion of Sulisobenzone taken by the formula:
308.31
C(2
V1 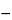 V2
V2),
in which 308.31 is the empirical weight of sulisobenzone;
C is the concentration, in moles per liter, of the 0.1 N tetrabutylammonium hydroxide VS; and
V1 and
V2 are the volumes, in mL, of titrant needed to reach the first and second endpoints, respectively.