Sulfate—
Dissolve 12.0 g in 37 mL of acetone, and add 3 mL of water. Titrate potentiometrically with 0.02 M lead perchlorate, prepared by dissolving 9.20 g of lead perchlorate in water to make 1000 mL of solution, using a pH meter capable of a minimum reproducibility of ±0.1 mV (see
pH  791
791
) equipped with an electrode system consisting of a lead-specific electrode and a silver-silver chloride reference glass-sleeved electrode containing a solution of tetraethylammonium perchlorate in glacial acetic acid (1 in 44): not more than 1.25 mL of 0.02 M lead perchlorate is consumed (0.02%).
Heavy metals—
Dissolve 1 g in 25 mL of acetone, and add 2 mL of water. Add 1.2 mL of thioacetamide-glycerin base TS and 2 mL of
pH 3.5 Acetate Buffer, and allow to stand for 5 minutes: any color produced is not darker than that of a control made with 25 mL of acetone and 2 mL of
Standard Lead Solution (see
Heavy Metals  231
231
) and treated in the same manner. The limit is 20 µg per g.
Related compounds—
Mobile phase—
Prepare a mixture of water, methanol, and glacial acetic acid (60:40:1). Make adjustments if necessary (see
System Suitability under
Chromatography  621
621
).
Diluent—
Prepare a mixture of methanol, water, and glacial acetic acid (70:30:4).
Reference solutions—
Prepare a solution in Diluent containing about 0.05 mg of 4-hydroxybenzoic acid and 0.025 mg of 4-hydroxyisophthalic acid per mL, and a second solution in Diluent containing about 0.05 mg of 4-hydroxybenzoic acid and 0.01 mg of phenol per mL.
Standard solution—
Dissolve accurately weighed quantities of 4-hydroxybenzoic acid, 4-hydroxyisophthalic acid, phenol, and the Salicylic Acid under test in Diluent to obtain a solution having known concentrations of about 0.05 mg per mL, 0.025 mg per mL, 0.01 mg per mL, and 50 mg per mL, respectively.
Test solution—
Prepare a solution of Salicylic Acid in Diluent containing 50 mg per mL. Sonicate until completely dissolved.
Chromatographic system
(see
Chromatography  621
621
)—The liquid chromatograph is equipped with a 270-nm detector and a 4.6-mm × 10-cm column containing 5-µm packing L1. The flow rate is about 0.5 mL per minute. Chromatograph the
Reference solutions, record the chromatograms, and note the retention times of the peaks. Chromatograph the
Standard solution, and record the peak responses as directed for
Procedure: the relative retention times are about 0.35, 0.45, 0.5, and 1.0 for 4-hydroxybenzoic acid, 4-hydroxyisophthalic acid, phenol, and salicylic acid, respectively, and the peaks are all baseline-resolved from each other. Changes in the composition of the
Mobile phase made to optimize resolution may change the elution order of impurities. Use the chromatograms obtained from the
Reference solutions to determine the elution order of the specified impurities.
Procedure—
Separately inject equal volumes (about 2 µL) of the
Standard solution and the
Test solution into the chromatograph, record the chromatograms, and measure the areas for the major peaks. Calculate the percentage of each relevant related compound taken by the formula:
2
Ci [
ri / (
rSi 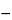 ri
ri)],
in which
Ci is the concentration, in mg per mL, of the relevant related compound in the
Standard solution; and
ri and
rSi are the peak responses for the relevant related compound obtained from the
Test solution and the
Standard solution, respectively: not more than 0.1% of 4-hydroxybenzoic acid, 0.05% of 4-hydroxyisophthalic acid, and 0.02% of phenol is found. Calculate the percentage of any other impurity, other than the solvent peak, observed in the chromatogram of the
Test solution by the same formula, except to use the concentration of 4-hydroxyisophthalic acid in the
Standard solution as
Ci; the response of the peak of 4-hydroxyisophthalic acid in the chromatogram obtained from the
Standard solution as
rSi; and the response of any other impurity as
ri: not more than 0.05% of any other impurity is found; and the sum of all of the related compounds and other impurities found is not more than 0.2%.
Assay—
Dissolve about 500 mg of Salicylic Acid, accurately weighed, in 25 mL of diluted alcohol that previously has been neutralized with 0.1 N sodium hydroxide, add
phenolphthalein TS, and titrate with 0.1 N sodium hydroxide VS. Each mL of 0.1 N sodium hydroxide is equivalent to 13.81 mg of C
7H
6O
3.